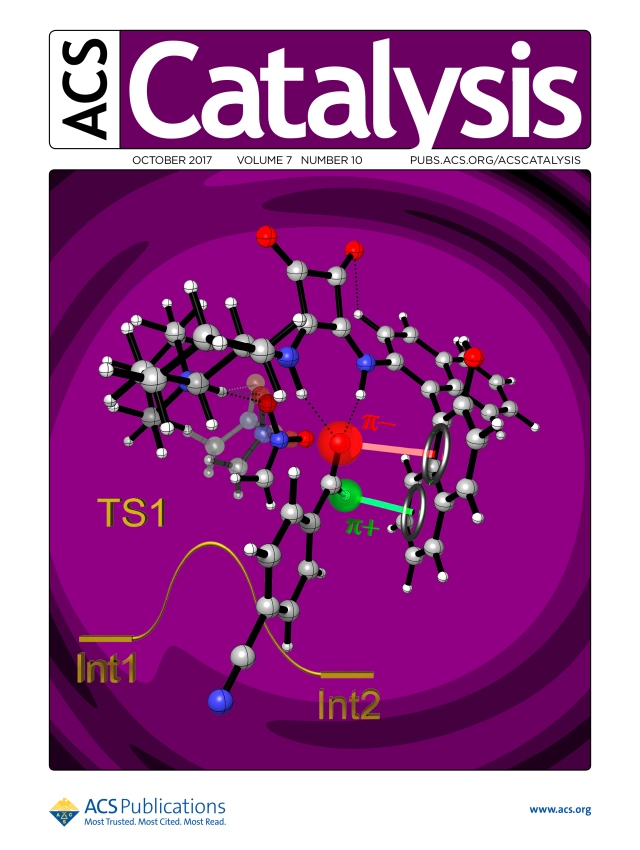
It is well known the lower activity of organocatalysts as compared to most metal catalysts. As a result, developing new and more potent organocatalysts focuses a great deal of interest both for science and chemical industry. Herein, we present our recent result aiming this strategic research line. In particular, we have designed, synthesised and tested a new squaramide which functions as a trifunctional organocatalyst when employed in the Henry reaction, as suggested by several studies carried out and described in this work.
Congratulations and thanks to the team, specially, this time, to Juanvi!
Trifunctional squaramide catalyst for efficient enantioselective Henry reaction activation. Alegre-Requena, J. V.; Marqués-López, E.; Herrera, R. P. Adv. Synth. Catal. 2016, Early view. DOI: 10.1002/adsc.201600046
Abstract. A new class of  trifunctional squaramide catalyst acting by means of multiple interactions has been found in a study of the Henry reaction. Enantiomerically enriched nitroaldol products were obtained in good yields and high enantioselectivities under mild conditions using one of the smallest amounts of organocatalyst reported so far for this reaction (0.25 mol%). The catalyst was able to generate hydrogen bonding and anion-π/hydrogen-π interactions with the substrates, responsible of the improvement in the reactivity and the enantioselectivity of this process. Computational calculations support a mechanistic hypothesis based on an anion-π effect, this being the first example reported in asymmetric catalysis.
trifunctional squaramide catalyst acting by means of multiple interactions has been found in a study of the Henry reaction. Enantiomerically enriched nitroaldol products were obtained in good yields and high enantioselectivities under mild conditions using one of the smallest amounts of organocatalyst reported so far for this reaction (0.25 mol%). The catalyst was able to generate hydrogen bonding and anion-π/hydrogen-π interactions with the substrates, responsible of the improvement in the reactivity and the enantioselectivity of this process. Computational calculations support a mechanistic hypothesis based on an anion-π effect, this being the first example reported in asymmetric catalysis.
Keywords: aldehydes; nitroalkanes; organocatalysis; squaramides; trifunctional catalysts; Henry reaction